The majority of electric cars use lithium-ion batteries, which are recharged by the movement of lithium ions between a positive electrode (cathode) and a negative electrode (anode). These batteries typically contain cobalt in the cathode, providing stability and high energy density.
Cobalt has drawbacks due to its limited availability and volatile price. Additionally, many of the countries with cobalt deposits are politically unstable. The extraction of cobalt also poses risks to workers and the environment, resulting in toxic waste that pollutes the surrounding land, air, and water.

As the demand for electric vehicles increases, the cost of cobalt batteries is expected to rise so MIT researchers have been working on finding alternative battery materials. Organic materials have been considered as a potential alternative to cobalt-containing batteries.
However, they have not been able to match the conductivity, storage capacity, and lifetime of these batteries. To improve conductivity, these materials are often mixed with binders like polymers, but this reduces the battery’s storage capacity as the binders make up a significant portion of the overall material.
Six years ago, Dincă’s lab started a project funded by Lamborghini to create an organic battery for electric cars. During their research on porous materials, they discovered a fully organic material that showed potential as a good conductor.
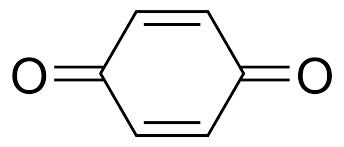
This material is made up of multiple layers of TAQ (bis-tetraaminobenzoquinone), a small organic molecule with three hexagonal rings. The layers can expand in all directions like graphite. The molecules contain quinones, which store electrons, and amines, which enable the material to create sturdy hydrogen bonds.
The hydrogen bonds in the material provide both stability and insolubility, which is crucial for preventing it from dissolving in the battery electrolyte and prolonging its lifespan, unlike certain organic battery materials. This can allow for over 2,000 charge cycles with minimal degradation.
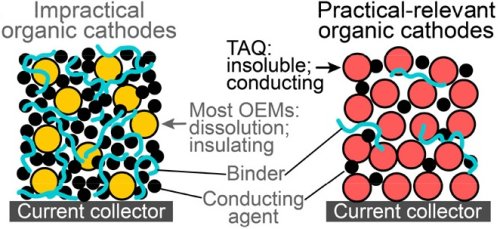
The material performed well in tests, with conductivity and storage capacity comparable to traditional cobalt-containing batteries. Additionally, batteries with a TAQ cathode can be charged and discharged more quickly than current batteries, potentially accelerating the charging time for electric vehicles.
The main materials required to make this type of cathode are readily available and widely produced chemicals called quinone precursor and amine precursor. The researchers believe that the cost of producing these organic batteries could be around 33% to 50% cheaper than cobalt batteries.
Lamborghini has obtained the rights to use the technology patented by Dincă’s lab.
Reference- Clean Technica, Interesting Engineering, Popular Science, Futurism, Inside EVs, Journal ACS Central Science, MIT News, Electrek






