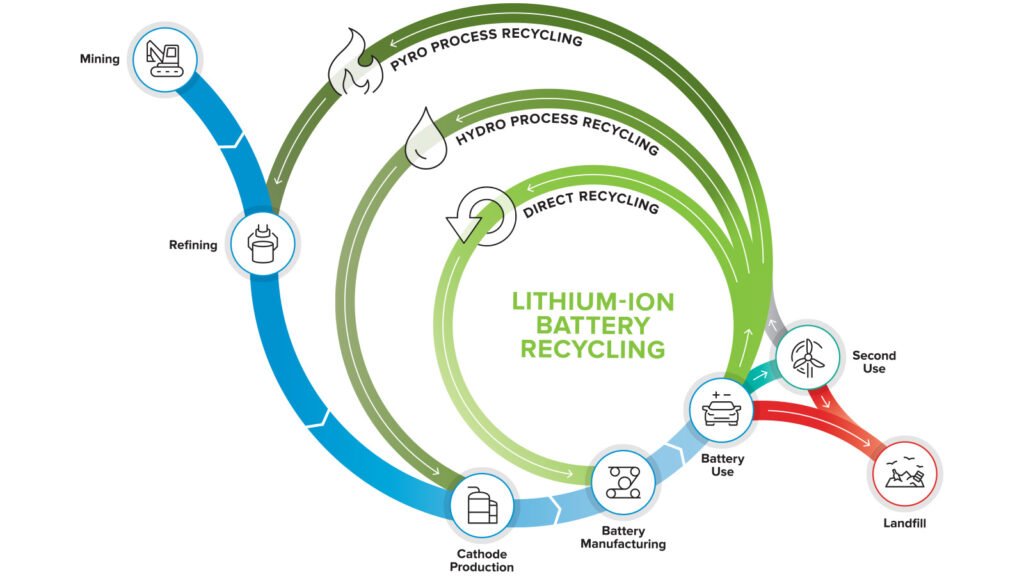
Lithium-ion batteries power our technology present and future. However, the growing usage of lithium-ion batteries, particularly in vehicles, has overtaken the technology for recycling them. In less than a decade, researchers project that two million tons of end-of-life lithium-ion batteries from EVs will be retired each year.
Scientists at the ReCell Facility have now made a crucial finding that removes one of the most significant barriers to making recycling lithium-ion batteries commercially viable.

Michigan Technological University (MTU) and ReCell researchers gave a new twist on an old process called froth flotation; a method which was used for separating individual cathode materials.
Used for many years by the mining industry to separate and purify ores, froth flotation separates materials in a flotation tank based on whether they repel water and float, or absorb water and sink. Generally cathode materials sink, which makes them difficult to separate from each other.
That’s true of lithium nickel manganese cobalt oxide (NMC111) and lithium manganese oxide (LMO), two common EV battery cathode materials that the ReCell team used in its experiments.
What the researchers found was that separation can be achieved by making one of the cathode materials, NMC111, float via the introduction of a chemical that makes the target material repel water.

After separating the cathode materials, the researchers established through testing that the method had no effect on the electrochemical performance of the materials. Both had excellent purity levels as well (95 percent or above).
The development of ReCell has far-reaching consequences, including lowering the cost of recycling lithium-ion batteries, fostering the creation of a lucrative recycling market for end-of-life lithium-ion batteries, and lowering the cost of EVs for both manufacturers and customers.
But, for the time being, the ReCell Center team is laser-focused on developing a comprehensive recycling process for lithium-ion batteries that is commercially viable, step by step. Only then will it become extensively used.
This a PR Newswire Feed; edited by Clean-Future Team






